|
Now that you know all the fundamentals related to ethyl alcohol or ethanol, let us examine how ethanol is obtained from ethene. In organic chemistry, the term alcohol refers to a compound containing the OH (hydroxy). From Ethene to Ethanol: Hydration of Ethene. Alcohol is a functional group that you are probably familiar with. The ‘ol’ of ethanol or ethyl alcohol is there because of this reason. carboxylic acid + alcohol and ester + water This is an acid-catalyzed equilibrium. Now, when we try to name the compounds that have this hydroxyl group, we use the suffix ‘ol’. Alcoholic compounds on reaction with ceric ammonium nitrate. Esters can be shown in text as: RCOOR or (occasionally) ROCOR. To identify the functional groups present in an organic compound. Hydroxyl is a functional group present in organic and inorganic compounds such as alcohols. EsterĮsters have a pair of alkyl or aromatic groups attached to a carbonyl + linking oxygen function. A functional group is a group of atoms that decide the properties and reactions of a particular molecule. 
The carbonyl group is an extremely function since numerous typical functional groups are based on a carbonyl, including aldehydes, ketones, carboxylic acids, esters, amides, acyl (acid) chlorides, acid anhydrides. Carboxylic acids can be displayed in the text as: RCOOH Carboxylic acids are weak Bronsted acids and they free CO2 from carbonates and hydrogen carbonates. (3) ALCOHOL: These compounds are third in the polarity because of hydrogen bonding capabilities and the presence of only one oxygen vs. Carboxylic acidĬarboxylic acids have alkyl or aromatic groups connected to a hydroxy-carbonyl function. Ketones give red-orange speeds up with 2,4-dinitrophenyl hydrazine. Ketones can be identified from aldehydes by providing negative test results with Fehling’s solution (brick red precipitate) or Tollens reagent (silver mirror). Hence the compound above is rightly named as 3-pen. Unlike the alkyl halides, this group has two reactive covalent bonds, the CO bond and the OH. Alcohol having higher priority than ENE should be chosen as the suffix. The functional group of the alcohols is the hydroxyl group, OH. Predict the molecular formula of hexanol.Ketones have a pair of alkyl or aromatic groups attached to a carbonyl function. IUPAC states that the higher priority functional group forms the suffix. 
They are a homologous series and have the general formula C n H 2n + 1 OH. So, 2 n + 1 = (2 × 10) + 1 = 20 + 1 = 21. Alcohol molecules all contain the hydroxyl (-OH) functional group. Predict the molecular formula of decanol and explain your answer. These beverages all contain one specific type of alcohol: ethanol. The general formula for the alcohols is C n H 2n+1 OH (where n is the number of carbon atoms in the molecule). If alcohol makes you think of wine, beer or liquor, you’re not too far off. Take care not to confuse the –OH group with the hydroxide ion, OH–. It is responsible for the typical reactions of alcohols. 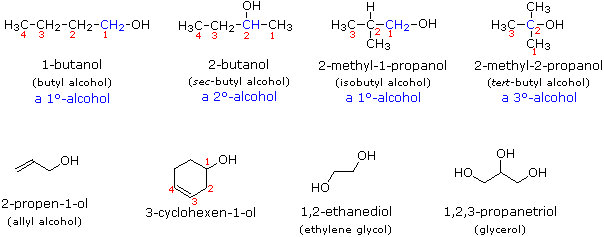
The functional group in the alcohols is the hydroxyl group, –OH. show a gradual variation in physical properties, such as their boiling points.differ by CH2 in molecular formulae from neighbouring compounds.Like all homologous series, the alcohols:
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
